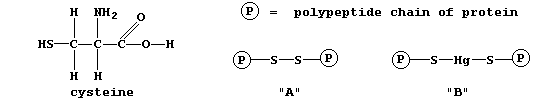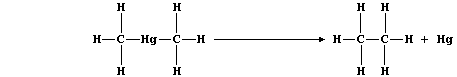METALS: MERCURY
Mercury, a very rare element in the Earth's crust (0.000008%), occurs
mainly in ores either as a sulfide or native. This metal, which has a
low melting point (-39ฐC) and a low boiling point (357ฐC), is a mobile
liquid at room temperature. Mercury's high density (13.6 g cm-ณ),
uniform coefficient of expansion, electrical conductivity and fluidity,
high volatility, and ability to dissolve many metals, has resulted in
its widespread use (e.g., in thermometers, as electrical contacts, in
vapour lamps, and in the formation of alloys known as amalgams).
[.. K > Ca > Na > Mg > Al > Zn > Fe > Sn > Pb > (H) > Cu > Hg > Ag ..]
1. Mercury is usually extracted by heating mercury(II) sulfide in air
to form mercury(II) oxide, followed by thermal decomposition of this
oxide to give mercury vapour (which is then condensed). Construct the
symbol equation for each reaction in this extraction process. _________
_______________________________________________________________________
_______________________________________________________________________
[4]
2. Mercury vapour is very dangerous because it destroys lung and brain
tissues. Suggest and explain one reason why areas affected by small
spillages of mercury are immediately covered with powdered sulfur. ____
_______________________________________________________________________
_______________________________________________________________________
[2]
3. The ingestion of small quantities of elemental mercury from dental
amalgams appears to result in no harmful effects. State and explain the
reason why mercury ions are not formed from mercury and the aqueous
hydrochloric acid present in gastric juice. ___________________________
_______________________________________________________________________
[2]
4. Presented here are partial explanations for the toxicity of, and
the tolerance shown towards, mercury compounds in living organisms ...
Proteins are formed by condensation reactions between carboxylic acid
(-COOH) and amino (-NH2) functional groups of amino acids. One of these
amino acids is known as cysteine, which contains a thiol (-SH) group.
Frequently, the thiol groups on the polypeptide chains react together
to form 'disulfide bridges' (i.e., as shown in "A"). These bridges are
important in determining the three-dimensional shapes of proteins and,
therefore, their correct biological function.
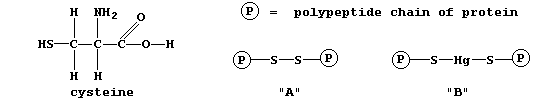
Mercury compounds react with these disulfide bridges to form proteins
containing inserted mercury (i.e., as shown in "B"); these insertions
change their (optimal) three-dimensional shapes, and so toxic effects
often result. By way of contrast, many living organisms have evolved a
limited degree of tolerance towards such compounds by biosynthesizing
'cysteine-rich' proteins, whose polypeptide chains contain numerous
thiol groups; these react similarly with mercury ions, and so act as an
effective homeostatic mechanism of detoxification.
(a) Name the group of catalytic proteins whose efficacy might decrease
as a result of mercury insertions into disulfide bridges. ____________
[1]
(b) Name another element whose ions might react with the thiol groups
of cysteine-rich proteins. ____________________________________________
[1]
5. Compounds of mercury are used, often indiscriminately, as biocides:
unfortunately, in common with those of a number of other heavy metals,
they are accumulated up the trophic levels. This diagram represents a
simple food chain in the complex Antarctic food web.

(a) The protoctistans Navicula perpusilla, apart from being the major
producers of chemical energy in marine food webs, provide a huge amount
of the biosphere's supply of dioxygen. By what light-dependent process
do these protoctistans produce this gas? ______________________________
[1]
(b) The crustaceans Euphausia superba, commonly known as 'krill', build
their exoskeletons by incorporating which metal ions? _________________
[1]
(c) Estimate the amount of mercury(II) ions which could be accumulated
by one blue whale (Sibaldus musculus), assuming that each protoctistan
absorbed 2 x 10-นณ g of mercury(II) ions and that none of the consumers
egest or excrete these ions. __________________________________________
_______________________________________________________________________
[2]
6. Certain anaerobic bacteria have evolved an ability to metabolize
elemental mercury, which is present in the environment largely because
of the illegal discharge of industrial waste. These organisms excrete
several toxic metabolites, including dimethylmercury; the biosynthesis
of this (lipid-soluble) compound involves vitamin-B12.
(a) Mercury's ability to form amalgams is utilized in the extraction of
precious metals - often in areas rightly considered as non-renewable
natural resources (e.g., rainforests). Name one precious metal which is
extracted via amalgamation with mercury. ______________________________
[1]
(b) Dimethylmercury thermally decomposes to give ethane and mercury;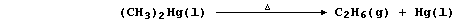
The equation for this thermal decomposition can be written as follows:
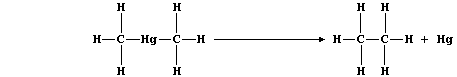
Complete the Table below using these bond energies (in kJ mol-น):
C-H (413); C-Hg (109); and C-C (346).
Bonds broken |
Energy absorbed
/ kJ mol-น |
Bonds formed |
Energy released
/ kJ mol-น |
|
|
|
|
|
|
|
|
Total = |
Total = |
[3]
Calculate the heat energy change (DH) for the above decomposition. ____
_______________________________________________________________________
[2]
(c) Hitherto, the metabolic activities of these bacteria have focused
attention on the implications for various ecosystems. Nevertheless, one
should also be aware that their use in a fermentation process could
provide an opportunity for developing a renewable energy source (i.e.,
ethane). Construct the symbol equation for the combustion of ethane.
_______________________________________________________________________
[2]
Dr. R. Peters Next Contents' List