ORGANIC CHEMISTRY: CATALYTIC HYDROGENATION
Margarine and butter are both mixtures of fats, and so are useful
sources of chemical energy in the diet. Margarine, however, is much
lower in cholesterol; this steroid is required for homeostasis, because
it is both an essential component of cell membranes and an intermediate
in the biosynthesis of the steroid hormones: but, consistently high
concentrations of cholesterol circulating in the bloodstream appear to
increase the chances of contracting cardio-vascular diseases.
1. Margarine is made from vegetable oils which contain unsaturated
carbons. Hydrogenation of these oils, at 170°C using a nickel catalyst,
saturates the carbon chains; this raises the melting point of the oils
so that they become solid at room temperature. Prompted by the need to
minimize energy costs, manufacturers have considered the possibilities
of biotechnology. Recently, one company's research team have isolated
several enzymes from anaerobic micro-organisms living in the Amazonian
rainforest (a non-renewable resource). In qualitative studies, one of
these enzymes was shown to be effective in the hydrogenation of a wide
range of unsaturated compounds; and in quantitative studies, controlled
experiments were used to determine the speed of these hydrogenations.
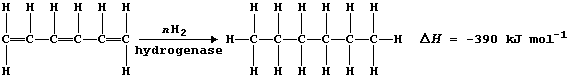
For example, the following hypothesis was examined: 'The speed (S) of
hydrogenating hexa-1,3,5-triene increases in direct proportion to the
amount (C) of hydrogenase enzyme "X"; i.e., S = k × C'; the Table shows
a summary of the chosen conditions and raw data.
Constants: hexa-1,3,5-triene (1.00 mole); volume of ethanol (1 dm³);
dihydrogen (excess); temperature (23°C); absence of light and dioxygen.
Amount (C) of "X"
hydrogenase / mmol |
0.00 |
0.50 |
1.00 |
1.00 |
1.00 |
1.50 |
2.00 |
Reaction time / s |
»3600 |
126 |
77 |
61 |
59 |
40 |
31 |
Reaction speed (S)
/ s-¹ |
0.000 |
|
|
|
|
|
|
(a) At room temperature (25°C) and pressure (100 kPa), what volume
of hydrogen gas is theoretically required for complete hydrogenation of
1.00 mole of hexa-1,3,5-triene? _______________________________________
[1]
(b) Suggest one reason why, in order to maintain a temperature of 23°C,
the reaction vessel needed to be externally cooled. ___________________
_______________________________________________________________________
[1]
(c) State the independent variable in the above investigation. ________
_______________________________________________________________________
[1]
(d) Calculate the missing values for the reaction speed, and insert
these data into the Table above.
[2]
(e) A value of a dependent variable which does not follow the general
pattern is referred to as an 'anomalous result'. In this investigation,
for example, the first reaction speed for C = 1.00 mmol appears
anomalous. Suggest one explanation for this anomaly. __________________
_______________________________________________________________________
[1]
(f) Label both axes, plot all seven data points, and then draw a best
straight line through as many points as is sensible.
_ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _
0.032_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
0.024_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
0.016_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
0.008_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
0.000_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
| | | | | | |
0 0.4 0.8 1.2 1.6 2.0 2.4
[4]
Determine the gradient of the graph; this value, 'k', is the
proportionality constant in the directly proportional relationship
S = k × C. (y2 - y1)
k = ————————— =
(x2 - x1)
[3]
Complete this precisely worded conclusion (which incorporates the
constants). 'The speed of hydrogenating 1.00 mole of hexa-1,3,5-triene
in 1 dm³ of ethanol at 23°C, in the absence of ______ and ________, is
directly proportional to the amount (C) of hydrogenase "X" (within the
range ________________); i.e., S = k × C, where k = _________________.'
[3]
Use the equation, and your value of k, to estimate the reaction time if
0.025 mmol of hydrogenase "X" was used. _______________________________
_______________________________________________________________________
[2]
(g) Carefully explain why the speed was slower, k = 0.0035 s-¹ mmol-¹,
when this series of experiments was repeated at 3°C. __________________
_______________________________________________________________________
_______________________________________________________________________
[2]
(h) In related series of experiments, the value of k increased with
increasing temperature upto 55°C - as expected. Suggest why, however,
the value of k was zero at all temperatures above 65°C. _______________
_______________________________________________________________________
[1]
(i) Recombinant DNA technology, popularly known as genetic engineering,
could be used to alter the genes of these anaerobic micro-organisms.
Suggest one desirable improvement in hydrogenase "X". _________________
_______________________________________________________________________
[1]
2. A significant fraction of the human body's cholesterol is used to
biosynthesize bile salts; these have detergent properties similar to
the salts of long-chain carboxylic acids, and so can act as emulsifying
agents for fats and oils. In which organ are bile salts produced? _____
_______________________________________________________________________
[1]
Dr. R. Peters Next Contents' List