ORGANIC CHEMISTRY: BIOCIDES
Chemical substances which kill living organisms are known as biocides.
[In Lewis Carroll's story, Through the Looking-Glass (London, 1872),
the Red Queen said: "Now, here, you see, it takes all the running you
can do, to keep in the same place."]
1. Various species of insects are regarded as pests because either
they act as vectors for pathogens (e.g., female Anopheles mosquitoes),
or they destroy crops (e.g., Locusta migratoria). Substances used for
destroying insect pests are known as insecticides, as typified by DDT.
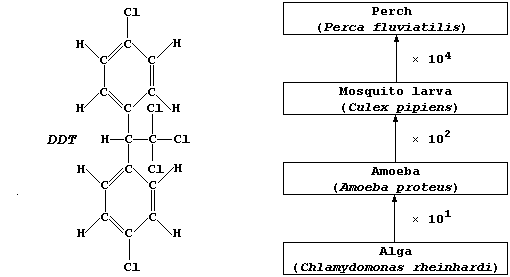
(a) DDT is non-selective, killing beneficial as well as pest species,
and accumulates up the trophic levels. The diagram above represents a
simple food chain in a typical freshwater lake. Estimate the amount of
DDT which could be accumulated by one perch, assuming that each alga
absorbed 1.0 µg and that none of the consumers egest or excrete DDT.
_______________________________________________________________________
[2]
Apart from predators and pollutants, state two factors which limit the
growth of algae. ______________________________________________________
[2]
(b) DDT's over-use has resulted in many short-lived organisms evolving
resistance; e.g., mosquitoes and tsetse flies (Glossina palpalis).
Thus malaria, which is caused by pathogenic protoctistans (Plasmodium),
has been controlled by the use of DDT on adult mosquitoes: but, ever
since DDT was first introduced in 1943, this method's effectiveness has
decreased, inevitably and inexorably, because populations of mosquitoes
resistant to DDT have evolved by the mechanism of natural selection.
Complete the following paragraph using the words from this list:
fittest; increased; inherited; meiosis; variation.
"Structural __________ has occurred in populations of Anopheles gambiae
mosquitoes due to natural mutations, followed by the exchange of genes
via ________ and random fertilization. Some of these mutations have
resulted in some mosquitoes containing dominant alleles which code
for the catabolism of DDT. These individuals have been the ________ in
environments where the agent of selection is DDT, and so more of these
have survived to reproductive maturity. Their offspring have __________
these favourable alleles; and so, within the gene pool of A. gambiae
mosquitoes, the frequency of DDT-resistant alleles has __________."
[5]
State one other method of destroying the vectors of malarial parasites
(Plasmodium), apart from using either pheromones or other insecticides
(e.g., malathion). ____________________________________________________
[1]
2. Bactericides include antiseptics; i.e., chemicals which, when used
at the correct concentration, kill pathogenic but not human cells. The
first antiseptic was 'Carbolic acid' (phenol), introduced by Lister in
1867, and two in household use today are 'TCP' (2,4,6-trichlorophenol)
and 'Dettol' (3,5-dimethyl-4-chlorophenol).
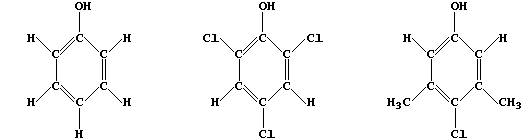
The order of bactericidal activity is: Dettol > TCP > Carbolic acid.
Study the substitution patterns and then, by completing the structural
formulae below, predict two compounds which might result in this order
of bactericidal activity: "U" > Dettol > TCP > "T" > Carbolic acid.
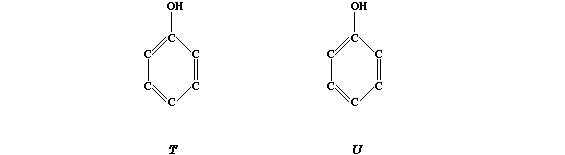
[2]
Apart from being effective in killing pathogenic bacteria, suggest two
desirable characteristics of a bactericide. ___________________________
_______________________________________________________________________
[2]
3. Plants use glucose, produced by photosynthesis, mainly in two ways:
first, as a respiratory substrate; and second, to synthesize all the
other compounds required for growth and reproduction [examples of these
'primary' metabolites include polysaccharides, nucleic acids, proteins,
and oils]. In order to discourage insect predators, many plants have
also evolved an ability to synthesize their own insecticides [examples
of these 'secondary' metabolites include caffeine, mustard, nicotine,
2-hydroxybenzenecarboxylic acid ("V"), and ethanedioic acid ("W")].
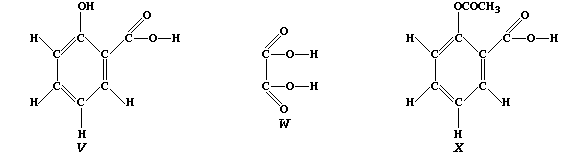
Compound "V", better known as salicylic acid, and structurally related
to aspirin ("X"), is used as an antiseptic and as a food preservative.
Because salicylic acid is only slightly soluble in water, manufacturers
prefer to use the water-soluble sodium salt. So as to summarize one
synthetic method for this salt, complete the following symbol equation:
C6H5OCOOH(aq) + NaHCO3(aq) —————————®
[2]
Dr. R. Peters Next Contents' List


