EXPERIMENTAL: AN INVESTIGATION INTO THE SPECIFIC HEAT CAPACITIES OF
WATER-GLYCEROL MIXTURES
Introduction
The physical and chemical properties of water differ from those of most
other liquids, but make it uniquely effective in supporting living
activities; e.g., water has the highest heat capacity of any liquid,
which means that it can absorb a lot of heat without the temperature
rising to levels which would denature cellular proteins.
However, water does have two properties which are certainly not optimal
for organisms living in certain environments. First, it freezes at 0°C:
so various animals which live in very cold habitats need some sort of
'anti-freeze', because the rate of diffusion is exceptionally slow in
frozen cells. And second, it has the highest water potential: so plants
which live in very salty habitats need a substance which will counter-
balance the low water potential of the external environment, otherwise
their cells would lose water by exo-osmosis. For some organisms (e.g.,
the algae Dunaliella), the biochemical solution to these differing
'problems' of water is to incorporate glycerol into their cytoplasm.
In this investigation, you are required to examine this hypothesis:
As the concentration (C) of glycerol increases, the specific heat
capacities (c) of the aqueous solutions decrease in direct
proportion; i.e., c = k × C. |
Method
1. Measure the mass of an insulated plastic cup; add about 150 cmł of
water; measure the mass of the cup and water; and, finally, subtract
these two masses to determine the mass of water (in kg).
2. Place a 12 V a.c. heater at a measured depth in the liquid, wait a
few minutes, and then record the starting temperature (in K).
3. The circuit for these experiments will already be constructed, so
close the switch and record the amount of energy (in kJ) required to
raise the temperature of the gently stirred liquid by 12 K; be careful
not to touch the heater with the thermometer.
4. Repeat steps 1 to 3 for each concentration of glycerol; i.e., 20%,
40%, 60%, and 80% (... and appropriate duplicates ...).
Notes
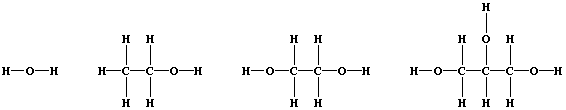
1. Shown below are the structural formulae of water, ethanol, ethane-
1,2-diol, and propane-1,2,3-triol (glycerol). These three alcohols are
miscible with water; their solubility is partially attributable to
'hydrogen bonding' [i.e., the weak electrostatic attraction between the
nucleus of a bonded hydrogen atom and a 'lone-pair' of electrons of a
bonded non-hydrogen atom (typically, nitrogen or oxygen)].
2. You will need to use this equation c = H ÷ (m × DT), where: c is
the specific heat capacity; H is the heat energy supplied; m is the
mass of substance; and DT is the temperature rise.
3. Your write-up should include, in addition to a complete summary of
results and calculations, a critical evaluation, a precisely worded
conclusion, and a suggested interpretation of the results.
Dr. R. Peters Next Contents' List
