SELECTED PRINCIPLES: INTRODUCTION - IONIC BONDING
Bonding between atoms, which occurs because the resulting molecule or
compound has a lower energy than its constituent atoms, is achieved by
redistributing the valence (or bonding) electrons. In ionic bonding,
this redistribution occurs by the atoms transferring one or more
electrons. The term ionic bond describes the electrostatic attraction
of two oppositely charged ions in a crystalline lattice.
The Mechanism of Ionic Bonding
Formally, the ionic compound sodium fluoride - which contains Na1+ and
F1- ions, each with an octet of electrons - results from the transfer
of one valence electron from a sodium atom to a fluorine atom; i.e.,
Na atom + F atom ——————————® Na1+ion + F1-ion
2,8,1 2,7 2,8 2,8
Despite the attractiveness of such a simple summary, one needs to peer
into the mechanism of ionic bonding in order to prevent misconceptions.
The formation of solid sodium fluoride, NaF(s), from solid sodium and
gaseous difluorine, can be divided into five ergonic processes; i.e.,
Na(s) ——————————® Na(g) DH1
Na(g) ——————————® Na1+(g) + 1e- DH2
F2(g) ——————————® 2F(g) DH3
F(g) + 1e- ——————————® F1-(g) DH4
Na1+(g) + F1-(g) ——————————® Na1+(s)F1-(s) DHL
Experiments show that formation of these gaseous ions is endothermic;
i.e., SDHI, the value of DH1 + DH2 + DH3 + DH4, is positive. In sharp
contrast, formation of the solid from these gaseous ions is exothermic;
i.e., the value of DHL, known as the lattice energy, is negative. Solid
sodium fluoride forms because the value of DH, DHL - SDHI, is negative;
so, the 'driving force behind' the formation of sodium fluoride is the
overwhelmimg exothermicity of the attractions of the oppositely charged
ions for each other (... and not the formation of ions with an octet).
Furthermore, because experiments have shown that, for every combination
of metallic and non-metallic element, the corresponding value of SDHI
is positive, the formation of an ionic compound is determined largely
by the value of DHL (the lattice energy).
This ('long-winded') approach above allows a qualitative understanding
of some apparent anomalies; two illustrative examples are given here.
Firstly, Na2O, MgO, Al2O3, NaCl, and MgCl2 are all ionic compounds in
which the ions have an octet: but, although aluminium and chlorine
can readily form ions with an octet, aluminium chloride is a covalent
compound. * The explanation is as follows: DHL - SDHI is negative in
each of the first five compounds, but DHL - SDHI is positive for the
hypothetical ionic compound Al3+(s)3Cl1-(s)
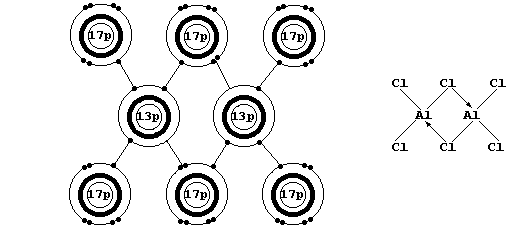
* When gaseous aluminium chloride is cooled, discrete AlCl3 molecules
dimerize to form Al2Cl6 (see the electron-structure diagram above).
And secondly, NaCl and MgCl2 are both ionic compounds in which the ions
have an octet: but, although transition elements do not form ions with
an octet, CuCl2 and FeCl2 are also ionic. The explanation is as before;
thus, DHL - SDHI is negative in each of these four compounds.
In the absence of either experimental or calculated data, the student
will ask (or even demand) to know how one predicts the type of bonding
in any given compound. Although there is no predictive method which is
foolproof, two rules of thumb are useful: first, the bonding between
non-metallic elements is normally covalent; and second, the bonding
between metallic and non-metallic elements is commonly ionic. *
The Structures and Physical Properties of Ionic Compounds
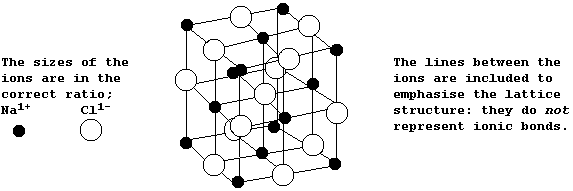
The diagram below shows the structure of NaCl; the oppositely charged
ions are arranged in a symmetrical pattern within a crystal lattice.
The electrostatic attractions between the oppositely charged ions in
the crystal lattices of ionic compounds are strong and omnidirectional:
and so, such compounds show several characteristic physical properties;
however, only three of these will be considered here.
First, they tend to have high melting points; a large amount of thermal
energy must be supplied to the crystal lattice before the ions vibrate
vigorously enough to overcome the electrostatic forces of attraction.
Second, they do not conduct electricity in the solid state, because
there are no free-moving ions to carry the current: contrastingly, they
do conduct electricity in either the liquid state or when dissolved in
water, because the ions are free to move.
And third, they tend to be insoluble in organic solvents and soluble in
water; the hydration energy released when ions are dissolved in water
is often similar to or greater than the lattice energy.
Not surprisingly, these characteristic physical properties are used as
evidence for ionic bonding: but there are at least three caveats.
First, high melting points are also observed for covalent substances
with giant structures [e.g., carbon-graphite and silicon(IV) oxide]. #
Second, the measurement of electrical conductivity can be precluded
because the compound either thermally decomposes before melting or has
a low solubility in water [e.g., many carbonates and nitrates decompose
before melting, and most oxides and sulfides are insoluble in water].
And third, a wide variety of covalent compounds are also soluble in
water [e.g., many alcohols and carbohydrates with small molar masses].
* In point of fact, the exceptions to this rule of thumb are legion.
However, such exceptions usually make no more than 'guest appearances'
in an introductory course (... a rare case of 'ignorance is bliss' ?).
# Typically, covalent substances with simple molecular structures have
low melting points, because the attractive forces between molecules are
weak, are insulators of electricity, because there are no free-moving
ions, and are soluble in organic solvents such as trichloroethane.
Dr. R. Peters Next Contents' List

