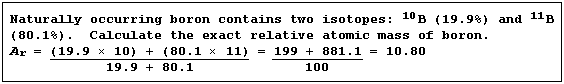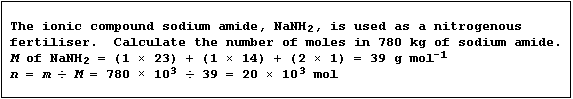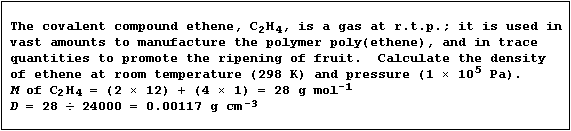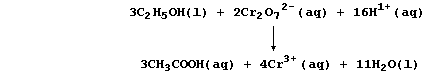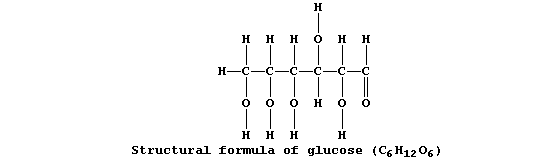AUFBAU1 [REFERENCE SECTION]
[01] INTRODUCTION - CHEMICAL CALCULATIONS (1)
[02] INTRODUCTION - CHEMICAL CALCULATIONS (2)
[03] INTRODUCTION - CHEMICAL EQUATIONS (1)
[04] INTRODUCTION - CHEMICAL EQUATIONS (2)
[05] ABUNDANCES / ALLOYS
[06] SALTS (FORMULAE / ANION & CATION TESTS)
[07] GASES (TESTS / LABORATORY PREPARATIONS)
[08] BIOLOGICAL TERMS
[09] NATURAL CYCLES
[10] POLLUTION
[11] INDUSTRIAL CHEMICALS
[12] REDOX POTENTIALS
REFERENCE SECTION: INTRODUCTION CHEMICAL CALCULATIONS (1)
Each generation of students is invariably treated to a version of the
following 'words of wisdom': "In my day, everybody knew their tables,
could spell correctly, and knew the capital of each country." Although
there is no evidence that Christopher Robin always went to Pooh-Bear's
tea-parties in those halcyon days of yore, it is worth accepting that
most fables do have grains of truth.
Certainly, it is true that calculations have proven to be the Achilles'
heel for a significant proportion of students. No investigation appears
to have been conducted which might cast light on the reasons for their
difficulties, but one can hazard the suggestion that such students may
have deferred the execution of calculations until late in their course.
Be that as it may, confidence in this topic can be acquired as follows:
beg, borrow, or buy a copy of J. A. Hunt and A. Sykes' well-structured
book (Chemistry Calculations, Longman, London, 1985); and then, from
the outset of the course, work steadily through it. The scope of the
text below is much more modest: primarily, the provision of a reference
set of examples of the commonest types of calculations.
Isotopes
A Periodic Table shows each element's atomic number; e.g., carbon has
the atomic number 6 (because each carbon atom contains 6 protons in its
nucleus). However, such a Table does not normally show that there is
usually more than one type of atom for each element; e.g., there are
three types of carbon atom: carbon-12 (¹²C), which contains 6 neutrons;
carbon-13 (¹³C), with 7 neutrons; and carbon-14, with 8 neutrons.
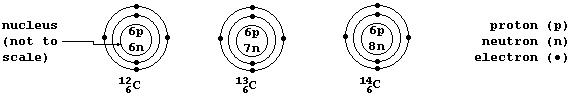
Isotopes are atoms of the same element that contain different numbers
of neutrons. So, C-12, C-13, and C-14 are the three isotopes of carbon.
The Mole
The mole is defined as 'the amount of substance that contains as many
elementary entities as there are atoms in exactly 12 g of the carbon-12
isotope'; these entities may be atoms, ions (charged atoms), molecules,
or other particles. Because the number of atoms in 12.00 g of carbon-12
has been determined experimentally as 6.022 × 10²³ (often referred to
as Avogadro's constant), one mole is equal to 6.022 × 10²³ particles.
The careful study of the Table below should prove rewarding.

* The seventh bear is, presumably, little more than a bit of fluff.
Relative Atomic Mass (Ar)
In Nature, elements usually exist as a mixture of isotopes. The (exact)
relative atomic mass of an element is a weighted mean of the relative
isotopic masses of the different isotopes; weighted, that is, in the
proportions in which they occur naturally.
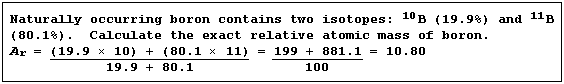
In some versions of the Periodic Table, the exact relative atomic mass
is given for each element; e.g., 35.4527 for chlorine. In others, each
value is approximated to the nearest whole number: though not that for
chlorine, which is shown as 35.5. There is absolutely no mathematical
or chemical justification for singling out chlorine; indeed, to be
brutally frank, the decision to do so was ill-judged. A trawl through
the exact masses of the elements reveals that roughly a quarter of them
warrant the appendage '.5', including nickel (58.69), copper (63.55),
zinc (65.39), gallium (69.72), and germanium (72.61).
Relative Molecular Mass (Mr)
The relative molecular mass (Mr) of a molecular element, of a covalent
compound, or of an ionic compound, is calculated by adding together the
(approximate) relative atomic masses in the molecular formula.

* Allotropes are two or more forms of the same element that differ in
both chemical and physical properties; e.g., three of the allotropes of
carbon are graphite, diamond, and buckminsterfullerene.
REFERENCE SECTION: INTRODUCTION - CHEMICAL CALCULATIONS (2)
Molar Mass (M)
The molar mass (M) of a substance has the same numerical value as its
relative atomic or molecular mass, but has the units g mol-¹.

Masses into Moles
The conversion of masses into moles requires the use of this equation:
Number of moles (n) of substance = Mass (m) ÷ Molar mass (M)
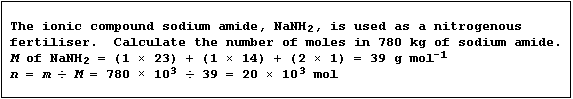
Moles into Masses
The conversion of moles into masses requires the use of this equation:
Mass (m) of substance = Number of moles (n) × Molar mass (M)

Concentration of a Solution
The concentration (C) of a solution, expressed as mol dm-³, is the
number of moles (n) of solute dissolved in a volume (V) of one cubic
decimetre of solvent; this quantity is calculated using this equation:
Conc. (C) of soln. = No. of moles (n) of solute ÷ Vol. (V) of solvent

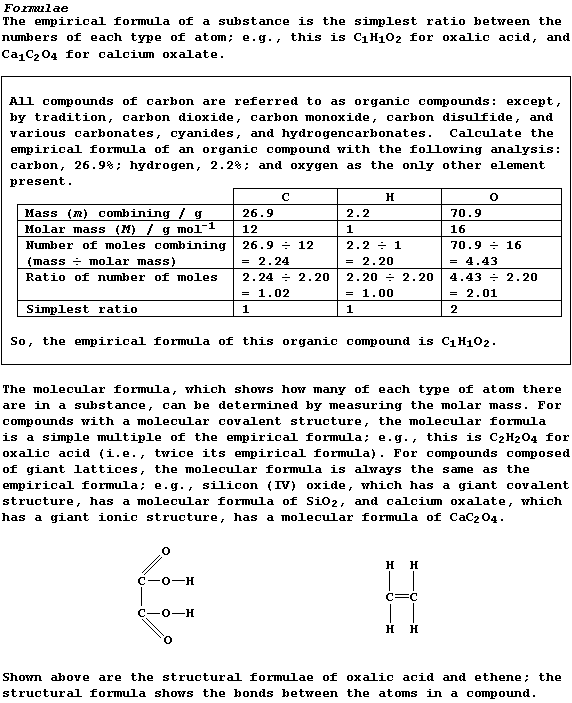
Density of a Gas
The density (D) of a gas, expressed in g cm-³, is the mass (m) of gas
contained in a volume (V) of one cubic centimetre. Because experiments
have shown that 1 mole of any gas, at room temperature and pressure
(r.t.p.), occupies a volume of 24000 cm³, the density of a gas can be
calculated using this equation:
Density (D) of a gas = Molar mass (M) ÷ 24000
REFERENCE SECTION: CHEMICAL EQUATIONS (1)
Chemical equations represent an essential part of the common language
of scientists. Accordingly, one needs to acquire the skills in both
their construction and interpretation. Setting aside the important fact
that all equations are derived from experiments, their construction
'simply' requires practice and patience using, wherever possible, model
examples: so, the text below focuses on the provision of a sound basis
for their interpretation.
A Chemical Equation
In a chemical reaction, the starting materials are known as reactants
and the substances formed as products. A chemical equation is used to
partially summarize a reaction. Thus, using chemical symbols, reactants
are placed on the left and products on the right of an arrow; and, to
conform with the Law of the Conservation of Mass, there must be the
same number of each type of atom on both sides of the equation; e.g.,
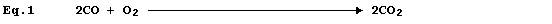
Equation 1, which summarizes the combustion of carbon monoxide, has two
equivalent meanings: first, 'two molecules of gaseous carbon monoxide
and one molecule of gaseous dioxygen react to give two molecules of
gaseous carbon dioxide'; and second, 'two moles of gaseous carbon
monoxide and one mole of gaseous dioxygen react to give two moles of
gaseous carbon dioxide'.
Equation 1 is absolutely complete, in the sense that nothing needs to
be added. Nevertheless, in attempts to provide more complete summaries
of reactions, scientists often festoon equations with a wide variety of
additional information; furthermore, equations are often replaced by
reaction schemes ...
State Symbols
Such symbols are abbreviations for the physical states of the reactants
and products; (aq), (g), (l), and (s) denote 'dissolved in water', gas,
liquid, and solid, respectively. In the absence of state symbols, the
reactants and products are assumed to be in their usual physical states
at room temperature (288 - 298 K) and pressure (100 kPa).
Equation 2 is merely equation 1 modified to include state symbols; and,
in this particular example, cannot be regarded as a useful improvement.
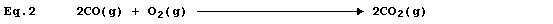
However, the absence of state symbols can result in errors. *
Equation 3 has adorned many textbooks, examination papers, and scripts.
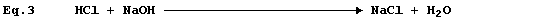
The precise interpretation of Equation 3 is, 'one mole of gaseous
hydrogen chloride and one mole of solid sodium hydroxide react to give
one mole of solid sodium chloride and one mole of liquid water': but,
under rigorously anhydrous conditions, such a chemical reaction would
be unlikely to occur; i.e.,

Equation 3 is frequently used to summarise the neutralization reaction
between aqueous solutions of hydrochloric acid and sodium hydroxide:
but the correct equation for this reaction is as follows.
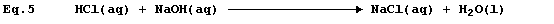
* The following anecdote may be illuminating. If there is one 'fact'
that people of un certain âge remember, from their best-forgotten days
studying Chemistry, it is that copper(II) sulfate is blue ...
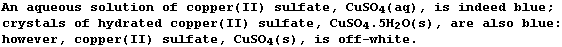
Energy Changes
Each chemical reaction is accompanied by the formation of one or more
new substances and an energy change (DE). Various symbols are used to
provide a qualitative (or quantitative) indication of such a change in
either a single reaction or a process, in particular: *
- DE, exergonic (i.e., energy is released to the surroundings);
+ DE, endergonic (i.e., energy is absorbed from the surroundings);
- DH, exothermic (i.e., heat energy is released to the surroundings);
+ DH, endothermic (i.e., heat energy is absorbed from the surroundings).
Reaction Conditions
Most chemical reactions are carried out under ambient conditions; i.e.,
under laboratory lighting at room temperature and pressure. Information
about reaction conditions, other than ambient, is placed on top of the
arrow; e.g., named catalyst, light, heat, low or high temperature, and
low or high pressure.
Reversible Reactions
In principle, each chemical reaction is reversible; i.e., it can occur
in both directions (and can be made to proceed in either direction by
altering the conditions): and so each equation should contain a double-
headed arrow. In practice, however, conditions are chosen which usually
ensure that there is complete conversion of reactant(s) to products(s);
and if this has been shown (or assumed) to be so, then such a reaction
is considered to be irreversible and is correctly summarized by an
equation which contains a single-headed arrow.
In industry, it is not always feasible to use conditions which ensure
irreversibility; thus, primarily for reasons of safety and economics,
many industrial-scale syntheses involve reversible reactions; e.g., ...
Nitrogen monoxide, an intermediate in the manufacture of nitric acid,
is obtained by the catalytic oxidation of ammonia; the symbol equation
for the industrial-scale synthesis of this gas is:

The meaning of this equation is as follows: 'at a temperature of 850°C
and a pressure of 200 kPa, in the presence of a platinum-rhodium
co-catalyst, the reaction of gaseous ammonia and dioxygen is reversible
(with incomplete conversion to gaseous nitrogen monoxide and water); in
a closed system, the forward and reverse reaction rates will be equal
at dynamic equilibrium'.
Reaction Schemes
A chemical equation is, strictly speaking, the only correct method of
summarizing a reaction. Nevertheless, in the scientific literature, the
appearances of such equations are fairly rare. One reason for their
rarity is that many symbol equations are quite complicated; e.g., the
oxidation of ethanol to ethanoic acid by acidified dichromate(VI) is:
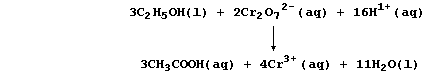
So, rather than bespatter their text with such 'monstrosities', authors
prefer to use a reaction scheme. In this 'chemical shorthand', only the
reactant and product of interest are stated explicitly; the reagent(s)
and conditions are placed over the arrow. So, the oxidation of ethanol
to ethanoic acid, for example, might be summarized by this scheme.
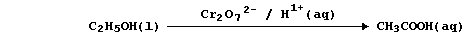
* In more advanced studies, the commonest symbol used to indicate the
energy change of a reaction is DG (i.e., the change in 'free energy').
REFERENCE SECTION: CHEMICAL EQUATIONS (2)
There are two pivotal moments in the development of a toddler: first,
when he or she realizes that 'Postie' probably does not hop on a sleigh
to the North Pole; and second, when he or she decides to collude with
adults in perpetuating the polite fiction of Father Christmas. Perish
the thought that examples of such fiction occur in Chemistry, but note
carefully the following sentence (J. Barrett, Understanding Inorganic
Chemistry, Ellis Horwood, Chichester, 1991): "At the school level, some
earlier theories are put forward as present-day explanations."
Perdre la grâce (ou Tomber en disgrâce)
Introductory courses invariably include a suite of experiments designed
to illuminate the important differences between elements, mixtures, and
compounds. By long-established tradition, the commonest suite involves
the following substances: the elements iron and sulfur; a mixture of
iron and sulfur; and the 'compound' iron(II) sulfide ...
A dark-grey solid is formed when a mixture of the above two elements is
gently heated; hitherto, this (fairly vigorous exothermic) process has
been summarized by the following equation.

This dark-grey solid, usually designated as the stoichiometric compound
iron(II) sulfide (FeS), has different chemical properties to both the
elements and the mixture; e.g., dilute hydrochloric acid reacts slowly
with iron to give the gas dihydrogen,
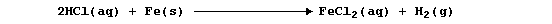
whereas the same acid reacts rapidly with the dark-grey solid to give
the (toxic) gas hydrogen sulfide,
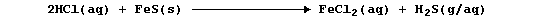
[Incidentally, this latter reaction is a splendid counterbalance to the
oft-stated paradigm 'acid + base gives salt + water only'.]
Chemists had known for decades that the designation of this dark-grey
solid as the stoichiometric compound iron(II) sulfide was not correct:
but, only in the 1980s did researchers show that substance obtained was
a mixture of phases (often called, rather loosely, 'non-stoichiometric'
compounds). No attempt is made here either to summarize this research *
or to explain its importance, simply because the science of phases is
so frightfully complicated: however, do note that a line is placed over
the nominal formula to indicate that a substance is non-stoichiometric,
and that chemical equations are modified; e.g.,

The 'fall from grace' of iron(II) sulfide as the archetypal compound
should, eventually, result in the removal of the suite mentioned above.
Surprisingly, perhaps, there are very few alternatives which fulfil the
two criteria of illumination and safety; of these, and by far the most
attractive, is a suite involving the following substances: the elements
zinc and iodine; a mixture of zinc and iodine; and the compounds water
and zinc iodide. To summarize the results of this suite might well be
regarded as profligate, but two cryptic 'cluettes' are surely in order.
The synthesis of aqueous zinc iodide is an exergonic process; i.e.,
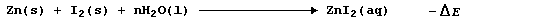
By contrast, but with a delightful feeling of logic, the electrolytic
decomposition of aqueous zinc iodide is an endergonic process; i.e.,
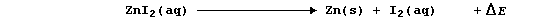
* See: J. K. Burdett and E. L. Miller, J. Am. Chem. Soc., 1987, 4081
(for a specific reference); F. A. Cotton and G. Wilkinson, Advanced
Inorganic Chemistry, Wiley, New York, 1988 (for a concise summary).
Cela sert à faire bouillir la marmite
Collectively, living organisms use a variety of respiratory substrates
as sources of chemical energy [including ethanol, glucose, and iron(II)
sulfide]; of these, the most extensively used is glucose:
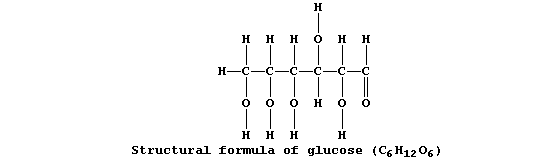
Equation 1, or its word equivalent, has been used in examination papers
and textbooks to summarize one fermentation process involving glucose.
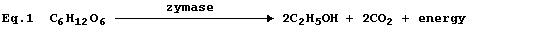
However, as written, equation 1 has little or no merit ...
First, and perhaps most importantly, equation 1 violates the Principle
of the Conservation of Energy - which states 'energy is neither created
nor destroyed, but can be transduced from one form to another'. All
reactions involve an energy change (DE): but energy is never produced.
Second, equation 1 is often - but, admittedly, not always - stated to
be a summary of the fermentation reaction: but this particular example
of fermentation summarizes a biological process that involves at least
eleven separate reactions.
Third, the inclusion of the word zymase is correct: but potentially
misleading. Thus, by analogy with amylase and catalase, amongst others,
one might reasonably assume that zymase is a single enzyme: whereas
zymase is a biological mixture, extracted from yeast (Saccharomyces),
that contains at least eleven enzymes.
And fourth, in the absence of state symbols, reactants are assumed to
be in their normal states at room temperature. In the solid state, the
rate of reaction between glucose and zymase would be immeasurably slow,
because there would be remarkably few collisions between the particles.
The above (rather brutal) deconstruction would be of precious little
value unless an acceptable alternative is available; happily, Equation
2 provides a most satisfactory summary of this fermentation process.
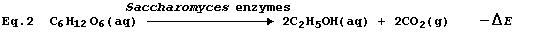
This fermentation process is but one example of respiration, whose
purpose in a living organism is to release energy in the usable form of
adenosine triphosphate (ATP). * Accordingly, the use of Equation 3 to
summarize this fermentation would be misleading, because it would imply
that the purpose of respiration is to release heat energy.
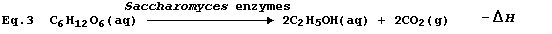
Nevertheless, in most types of respiration, it is true that about 60%
of the energy transduced (DE) is released as heat energy. #
* ATP, which is the immediate source of chemical energy for all living
organisms, provides each living cell with the energy required for its
endergonic processes (e.g., active transport and active transport).
# During their (short-lived) flowering period, Arum lilies carry out a
an alternative respiration process in which all the energy is released
as heat; reproduction is clearly an «energy sapping affair».
REFERENCE SECTION: ABUNDANCES
Most elements occur in the Earth's crust in a chemically combined state
as minerals; a mineral is a naturally occurring pure compound, whereas
an ore contains one or more minerals mixed with other substances [such
as sand, which is mainly silicon(IV) oxide]. The two commonest elements
in the Earth's crust are the non-metals oxygen and silicon (46.4% and
28.2%, respectively); eight of the next ten commonest are metals (see,
The Handbook of Physics and Chemistry, CRC Press, Boston, 1990). * #
First, complete the Table below, which lists the abundances of the
eight commonest metals in the Earth's crust, by inserting the correct
name of the metal (note the formula and Mr of the stated compound).
Metal (M) |
Most abundant compound of metal M |
Name |
Abundance / % |
Formula |
Mr |
Aluminium |
8.3 |
M2O3 |
102 |
|
5.6 |
M2O3 |
160 |
|
4.2 |
MCO3 |
100 |
|
2.4 |
MCl |
58.5 |
|
2.3 |
MCO3 |
84 |
|
2.1 |
MCl |
74.5 |
|
0.6 |
MO2 |
80 |
|
0.1 |
MO2 |
87 |
[7]
And second, noting that the qualitative independent variable is shown
as separate blocks on the horizontal axis, and that the quantitative
dependent variable is a linear scale on the vertical axis, complete the
bar-graph of the data above.
_ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _
A |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
b 8.0_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
u |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
n |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
d |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
a |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
n 6.0_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
c |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
e |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
o |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
f 4.0_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
m |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
e |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
t |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
a 2.0_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
l |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
/ |_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
% 0.0_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|_|
| Al | | | | | | | | | | | | | | |
Metal (M)
[7]
* The Earth's crust is regarded as the layer measured from the surface
to a depth of about 40 km.
# Hydrogen and carbon, the elements which form more compounds than all
the others together, are the 10th and 17th most abundant, respectively.
REFERENCE SECTION: ALLOYS
An alloy can be defined as 'a solid solution composed of two or more
metals, or of a metal or metals with one or more non-metals'. Although
alloys may be used either to replace or to conserve more expensive
metals, most are used to change the metals' physical properties, (i.e.,
conductivity, density, ductility, hardness, lustre, malleability,
melting point, and tensile strength), and/or chemical properties (e.g.,
resistance to corrosion). *
Complete the Table below, which includes some of the most important
alloys, by inserting the correct name of the alloy from this list:
Brass; Bronze; Cupro-nickel; Dental amalgam; Duralumin; Manganin;
Nichrome; Pewter; Solder; Stainless steel; and Steel.
Alloy |
Typical composition |
Special properties |
|
Fe(99%), C(1%) |
Stronger than iron, and more
corrosion resistant |
|
Al(95%), Cu(4%),
Mn(0.5%), Mg(0.5%) |
Density as low as aluminium,
but much stronger and more
corrosion resistant |
|
Cu(85%), Sn(15%) |
Stronger than copper, but just
as corrosion resistant |
|
Fe(85%), Cr(14%),
Ni(1%) |
Harder than steel, and much
more corrosion resistant |
|
Sn(85%), Cu(7%),
Bi(6%), Sb(2%) |
Stronger than tin, but still
easy to etch and engrave |
|
Cu(81%), Mn(15%),
Ni(4%) |
Suitable for fixed resistors
because of high resistivity and
low temperature coefficient |
|
Cr(80%), Ni(20%) |
Suitable for resistance heating
because of high resistivity and
high melting point |
|
Cu(75%), Ni(25%) |
Attractive appearance for coins
(looks like silver) |
|
Pb(67%), Sn(33%) |
Harder than lead, but has a
much lower melting point |
|
Cu(60%), Zn(40%) |
More easily shaped by stamping
and machining than bronze |
|
Sn(44%), Hg(33%),
Ag(22%) |
Resistant to corrosion from
the acidic products excreted
by mouth bacteria |
[11]
* An alloy should not be defined as 'a mixture of metals ...'. Whilst
an alloy is indeed a mixture, the term 'mixture' implies that there is
no chemical bonding between the components: whereas, usually, there is
between these in solutions. To place this ostensibly subtle difference
in true perspective, compare or reflect upon the physical and chemical
properties of the contents of two test-tubes: one containing a mixture
of powdered tin and lead, and the other solder.
REFERENCE SECTION: FORMULAE and SOLUBILITIES of SALTS
Salts are defined as 'ionic compounds which contain cations other than
hydrogen and anions other than oxide or hydroxide'. However, salts may
be more usefully considered as ionic compounds in which the ionizable
hydrogen ions of an acid have been replaced by either metal or ammonium
ions. A monoprotic acid, such as ethanoic acid, has one ionizable
hydrogen, and so can only form a neutral salt: by contrast, a diprotic
acid, such as carbonic acid, has two ionizable hydrogens, and so forms
both neutral and acid salts. In general, neutral salts are less soluble
in water but more thermally stable than their acid analogues; compare,
for examples, sodium carbonate with sodium hydrogencarbonate or calcium
carbonate with calcium hydrogencarbonate.
Complete the Table below, which summarizes the solubilities in water
and the formulae of typical neutral salts (formally) derived from five
acids [HCl(aq) ; H2SO4(aq) ; CH3COOH(aq) ; H2CO3(aq) ; and HNO3(aq)],
by inserting the correct formula for each salt. * # §
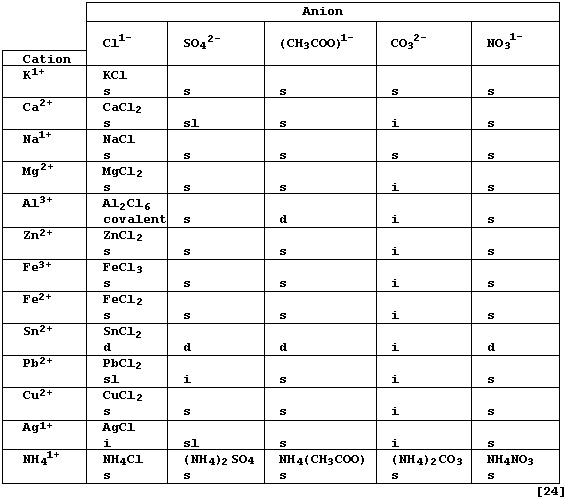
* Each formula refers to the anhydrous salt, although many of these
compounds crystallize from aqueous solutions as hydrates; e.g., calcium
chloride-water (1/6). The anhydrous salt can sometimes be obtained from
from the hydrated salt either by the use of certain dehydrating agents
(e.g., concentrated sulfuric acid), or by careful heating (though this
is often accompanied by thermal decomposition of the anhydrous salt).
# Solubility in water at 20°C: d = decomposes (i.e., hydrolyzes); i =
insoluble (< 1 g kg-¹); sl = slightly soluble (1 - 10 g kg-¹); and s =
soluble (> 10 g kg-¹).
§ Ionic compounds, which tend to have high melting points, conduct
electricity when their ions are free to move (i.e., in the molten state
or when dissolved in water: but not in the solid state).
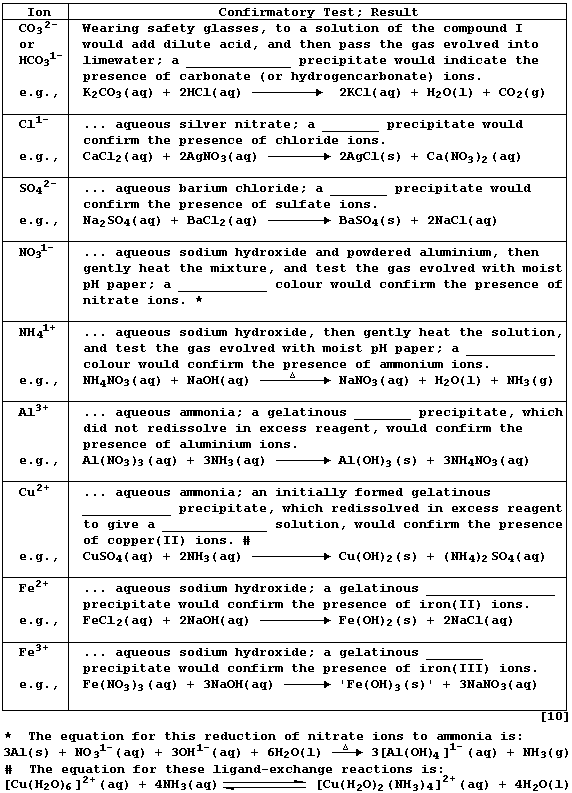
REFERENCE SECTION: TESTS for ANIONS and CATIONS
Confirmatory tests for ions share two common features. First, each test
is executed on a solution of the compound; this is usually prepared by
dissolving the solid in either distilled water or dilute nitric acid.
And second, reaction of an aqueous solution of the ion with the test
reagent results in either the selective precipitation of the ion or the
selective evolution of a characteristic gas.
Complete the Table below, which summarizes tests for ten common ions,
by inserting the correct observation from this list: brown; dark-blue;
milky-white; pale-blue; royal-blue; very pale-green; white.
REFERENCE SECTION: TESTS for GASES
Complete the Table below, which summarizes tests for ten common gases,
by inserting the correct observation from this list: bleaches; blue;
brown; dark-blue; decolourizes; green; milky-white; pink; 'pop'; red;
relights; white.
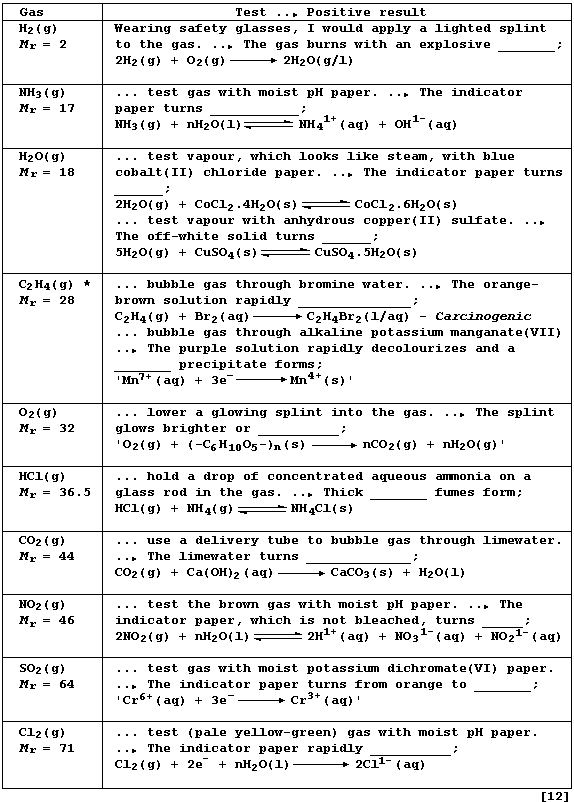
* Liquid and solid alkenes are immiscible with bromine water and with
alkaline potassium manganate(VII); accordingly, they require warming or
shaking. Note that alkanes also react with bromine: but such reactions
are very slow indeed - particularly in the absence of light - so the
orange-brown colour of bromine water usually remains.
REFERENCE SECTION: LABORATORY PREPARATIONS of GASES
Complete the Table below, which summarizes the laboratory preparations
of six common gases, by completing the balanced symbol equations.
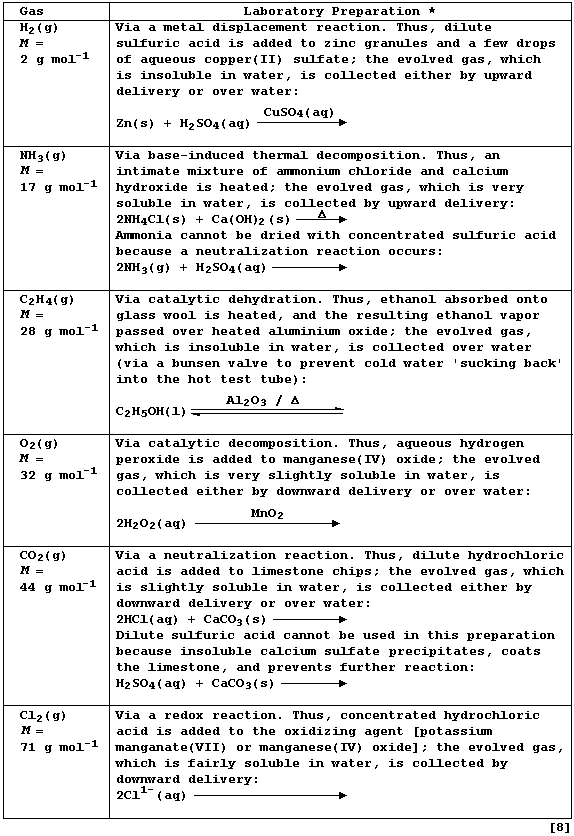
* Each preparation is routinely executed, in a well-ventilated fume
cupboard, wearing safety glasses. Most gases can be dried by passage
through calcium chloride, silica gel, or concentrated sulfuric acid;
however, the corrosive nature of the latter drying agent introduces a
unnecessary safety hazard.
REFERENCE SECTION: BIOLOGICAL TERMS
Complete the Table below, which defines or describes some important
biological terms that are occasionally included in chemistry texts, by
inserting the correct word from this list: Active transport; Allele;
Autotroph; Cell; Diffusion; Codominance; Dominant allele; Enzyme;
Excretion; Food chain; Gene; Genotype; Growth; Herbivores & Carnivores;
Heterotroph; Heterozygous; Homeostasis; Homozygous; Irritability;
Limiting factor; Movement; Nutrition; Organism; Osmosis; Phenotype;
Population; Recessive allele; Reproduction; Respiration; Saprotrophs;
Sex-linked allele; Species; Trophic level.
Term |
Definition or Description |
|
A structure containing the genetic code for
reproduction. |
|
A group of organisms capable of breeding to produce
fertile offspring; e.g., Ursos arctos (brown bear). |
|
A group of individuals in a species that live in a
defined habitat. |
|
An assembly of fragile molecules, enclosed within a
semi-permeable membrane, which show some or all the
other characteristics of living things. * |
|
The maintenance of a constant internal environment
despite changes in the external environment. |
|
The removal of waste products of cellular metabolism. |
|
The change in position of whole or part of a living
organism. |
|
The enzyme-controlled release of energy, in the form
of ATP, by the oxidation of organic compounds. |
|
The formation of new organisms to ensure the survival
of the species. |
|
The ability to detect and respond to changes in the
external environment. |
|
The processes by which chemical energy is obtained
and assimilated. |
|
The irreversible increase in dry mass of an organism
via biosynthesis and mitotic cell division. |
|
A catalytic protein. |
|
The net movement of particles down the concentration
gradient; i.e., from high to low concentration. |
|
The net movement of solute particles, across a
semi-permeable membrane, against the concentration
gradient; i.e., from low to high concentration. |
|
The net movement of water particles, across a semi-
permeable membrane, down the concentration gradient;
i.e., from high to low concentration. |
Term |
Definition or Description |
|
An organism which biosynthesizes its chemical energy
from inorganic compounds. |
|
An organism which obtains its chemical energy from
the organic compounds produced by other organisms. |
|
Ingestive heterotrophs which obtain their chemical
energy from living organisms. |
|
Absorptive heterotrophs which obtain their chemical
energy from dead organisms and their excretions. |
|
A series of organisms through which chemical energy
is transferred by feeding. # |
|
The position of an organism in a food pyramid. |
|
Any variable which reduces the rate of a biological
process from its possible maximum. |
|
The basic unit of inheritance; in molecular terms,
it is the sequence of DNA nucleotides which codes for
the synthesis of one polypeptide. $ |
|
One of a number of alternative forms of a gene. |
|
The allele(s) present for a specific characteristic. |
|
The physical or chemical expression of the genotype. |
|
One allele of a pair which has an effect on the
phenotype when homozygous or heterozygous. (Shown in
a genetic diagram by a capital letter; e.g., H.) |
|
One allele of a pair which has an effect on the
phenotype when homozygous. (Shown in a genetic
diagram by a small letter; e.g., h.) |
|
When both alleles in a heterozygous individual are
expressed in the organism's phenotype. |
|
An alternative form of a gene which is carried on a
sex chromsome. |
|
Where two alleles for a characteristic are the same
(e.g., HH or hh). |
|
Where two alleles for a characteristic are different
(e.g., Hh). |
[33]
* Most living organisms usually show nine characteristics; i.e.,
cellular organisation, homeostasis, excretion, movement, respiration,
reproduction, irritability, nutrition, and growth. (... 'Chemrring' ?).
[Viruses, which are invariably parasitic and pathogenic, are viewed as
non-living organisms because they do not show these characteristics.]
# A similar transfer occurs in a food web, but involves interacting
organisms (i.e., communities).
§ Despite their surprisingly frequent appearances, the terms 'dominant
gene' and 'recessive gene' are both incorrect.
REFERENCE SECTION: NATURAL CYCLES
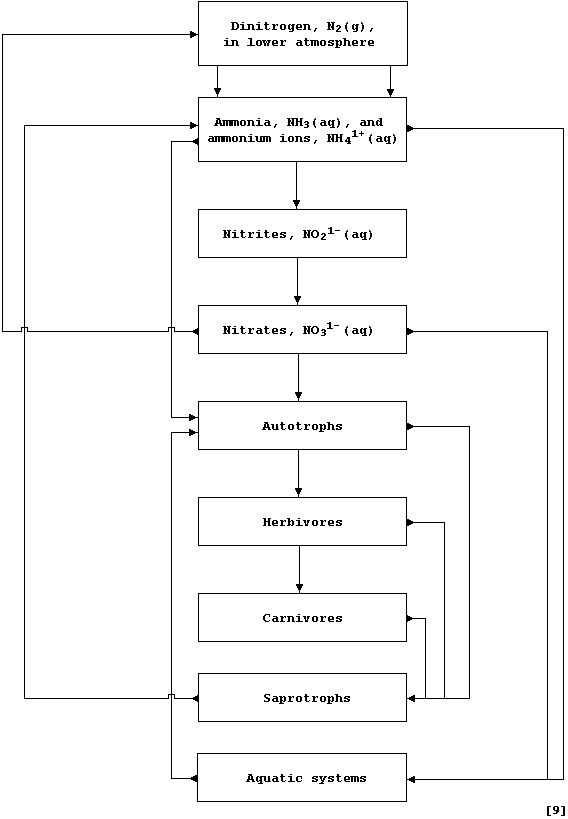
The biogeochemical recycling of nitrogen, carbon, and water are three
important examples of matter being continually recycled in Nature. *
Complete this first diagram, which is a simplified summary of the
Nitrogen cycle, by inserting the name of the correct process from this
list: Absorption; Biological fixation; Death; Decay; Denitrification;
Electro- and Photo-chemical fixation; Feeding; Leaching; Nitrification.
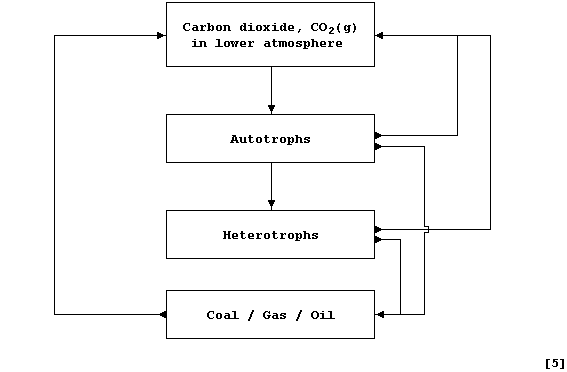
Complete this second diagram, which is a simplified summary of the
Carbon cycle, by inserting the name of the correct process from this
list: Combustion; Decomposition (anaerobic); Feeding; Photosynthesis;
Respiration.
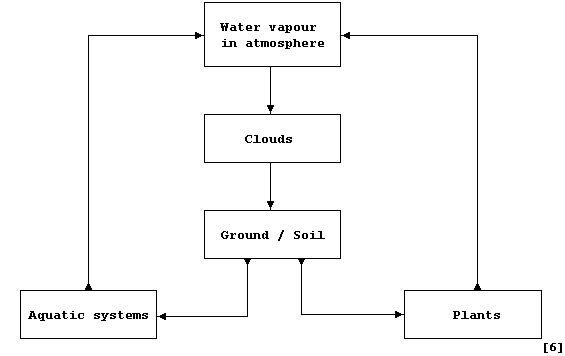
Complete this third diagram, which is a simplified summary of the
Water cycle, by inserting the name of the correct process from this
list: Absorption; Condensation; Drainage; Evaporation; Precipitation;
Transpiration.
* Ecology is the study of how organisms interact with their physical
and biological environments; and how these interactions influence both
the distribution and abundance of living organisms: in practice, since
the Industrial Revolution, the greatest influence has been Man.
REFERENCE SECTION: POLLUTION
The root cause of pollution is Man's unique ability to over-use the
biosphere's natural resources; 45% is a conservative guesstimate of
this use by just one species. Although most visible on ingestive
heterotrophs, particularly vertebrates, it is the effects of pollution
on autotrophs and saprotrophs that is important; this is because most
pollutants interfere with food webs by either increasing the natural
mutation rates or decreasing the reproductive rates of these organisms.
Complete the Table below, which summarizes the sources of the most
important pollutants and their biological and/or ecological importance,
by inserting the correct name of the pollutant(s) from this list:
Acidic oxides (e.g., NO2, SO2, and SO3); Biocides (e.g., Warfarin and
DDT); Carbon dioxide; Carbon monoxide; Chlorofluorocarbons (CFCs);
Hot water discharges; Lead compounds; Metal ions [e.g., Hg2+(aq) and
Cd2+(aq)]; Methane; Nitrogen oxides; Non-biodegradable detergents;
Phosphates and nitrates; Radioactive emissions (a- and b-particles and
g-rays); Sewage; Unburned hydrocarbons.
Source(s) |
Pollutant(s) |
Biological/Ecological Importance |
Vehicle exhausts
Paints
Mine workings
Storage batteries |
|
Accumulate up trophic levels.
Inhibit enzymes. |
Mine-workings
Industry |
|
Accumulate up trophic levels.
Inhibit enzymes. |
Nuclear power
stations |
|
Cause mutations; i.e., changes in
the sequence of nucleotides in
deoxyribonucleic acid (DNA) or in
ribonucleic acid (RNA). |
Refrigerants and
Aerosols
Vehicle and jet
exhausts
Agriculture and
Landfill sites |
|
Deplete ozone layer, allowing more
u.v. light to reach biosphere: so
increased mutation rates, as well
as decreased photosynthesis by
marine protoctistans. Contribute
to 'greenhouse effect'. * |
Fossil fuel power
stations
Vehicle exhausts
'Slash and burn'
agriculture |
|
Contributes to 'greenhouse effect'.
May increase plant yields, because
the gas is a limiting factor in
photosynthesis, and (indirectly)
increase the number of insects. |
Power stations |
|
Denature the enzymes of aquatic
organisms. Decrease concentrations,
in aquatic habitats, of dissolved
gases (e.g., O2, CO2, and N2). |
Fossil fuel power
stations
Vehicle exhausts |
|
Reacts reversibly but strongly with
haemoglobin, and so reduces
the availability of dioxygen to
aerobically respiring cells. |
Households
Factories |
|
Cause 'foaming', and so prevents
the diffusion of gases essential to
aquatic organisms. |
Source(s) |
Pollutant(s) |
Biological/Ecological Importance |
Households
Factories |
|
Provides rich source of chemical
energy for pathogens to reproduce.
Cause eutrophication. # |
Agriculture;
NPK fertilizers |
|
Cause eutrophication. Contribute to
soil erosion. Nitrates are soluble,
so maybe present in water supply. |
Agriculture
Forestry
Human habitats |
|
Accumulate up trophic levels. Can
cause mutations. Are not always
selective to pests, which usually
evolve resistance rapidly. $ |
Vehicle exhausts |
|
Contribute to photochemical smog,
which destroys various biological
molecules, decreases efficiency of
gas-exchange surfaces in animals,
and increases mutation rates. |
Coal-fired power
stations |
|
Dissolve in atmospheric water
vapour, which precipitates as 'acid
rain'; the acidic environments
result in the release of toxic ions
[e.g., aq. Al(III) and Pb(II)], as
well as reduced reproductive rates.
Contribute to photochemical smog. |
[15]
* The 'greenhouse effect' is caused by certain gases in the atmosphere
trapping heat radiated from the Earth. These 'greenhouse' gases absorb
outgoing infra-red radiation but do not significantly affect incoming
visible radiation (i.e., they act like panes of glass in a greenhouse);
the higher their concentration, the more radiation returned and so the
higher the temperature of the Earth's surfaces. 'Global warming' is the
gradual increase in temperature of the lower atmosphere, as a result of
the steady accumulation of greenhouse gases; its effects are expected
to include rises in sea levels, decreased amounts of moisture in soils,
milder winters, and disruption of delicately balanced ecosystems.
# The consequences of eutrophication, or over-enrichment, of water are
as follows. Nutrient ions are one of the limiting factors in the growth
of algae in aquatic habitats: so, these photosynthetic protoctistans
reproduce rapidly when these ions are in abundance. The phase of rapid
reproduction is followed by death of the entire population of algae,
because of the decreased availability of limiting factors (i.e., ions,
carbon dioxide, or light); the dead algae, which often release toxins,
are then decomposed by saprotrophic bacteria, whose aerobic activities
reduce a habitat's dioxygen supply to levels which cannot support other
aerobic organisms (e.g., crustaceans and fish).
§ The evolution of pest populations resistant to a biocide occurs via
the mechanism of natural selection, as exemplified by the evolution of
brown rats (Rattus norvegicus) resistant to Warfarin (a blood-clotting
toxin). Thus, structural variation has occurred in rat populations due
to natural mutations, followed by the exchange of genes via meiosis and
random fertilization. Some of these mutations have resulted in some
rats containing alleles which code for the catabolism of Warfarin.
These individuals have been the fittest in environments where the agent
of selection has been Warfarin, and so more of these have survived to
reproductive maturity. Their offspring have inherited these favourable
alleles; and so, within the gene pool of brown rats, the frequency of
Warfarin-resistant alleles has increased.
REFERENCE SECTION: USES of INDUSTRIAL INORGANIC CHEMICALS
Chemical and Engineering News, a publication of the American Chemical
Society, provides annual rankings of the top 50 industrial chemicals
produced in the United States; 12 of the top 18 are inorganic, whereas
the remaining 6 are organic.* A notable absence from these rankings is
dihydrogen; although produced in vast quantities, this gas is usually
used in the industrial plant where it is generated.
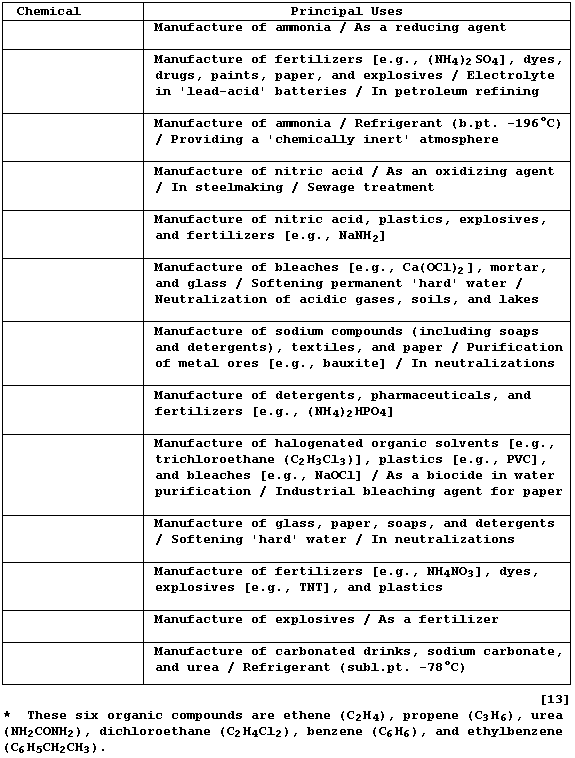
Complete the Table below, which summarizes the principal uses of the
top 13 industrial inorganic chemicals produced, by inserting the name
of the correct chemical from this list: Ammonia; Ammonium nitrate;
Carbon dioxide; Dichlorine; Dihydrogen; Dinitrogen; Dioxygen; Lime
(Calcium oxide and Calcium hydroxide); Nitric acid; Phosphoric acid;
Sodium carbonate; Sodium hydroxide; Sulfuric acid.
REFERENCE SECTION: MANUFACTURE of INDUSTRIAL INORGANIC CHEMICALS
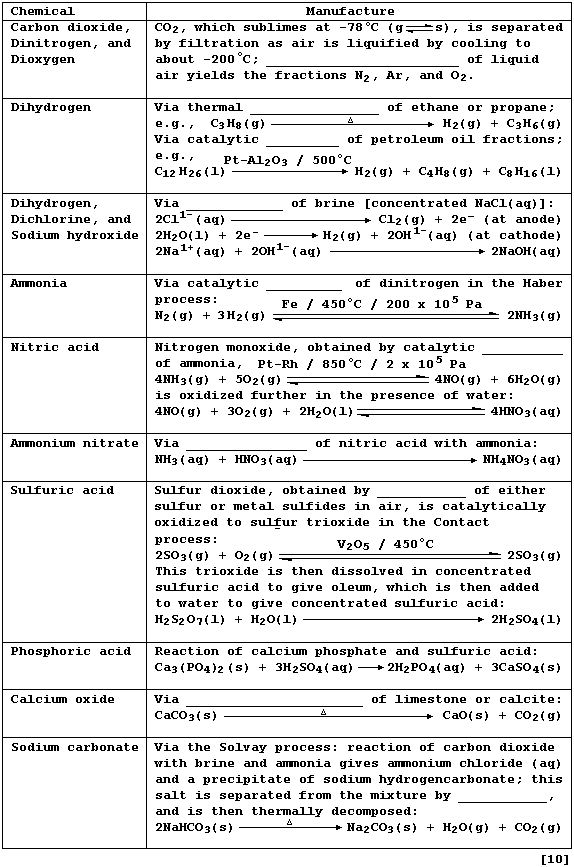
Complete the Table below, which summarizes the manufacture of the top
industrial inorganic chemicals produced, by inserting the name of the
correct process from this list: combustion; cracking; dehydrogenation;
electrolysis; filtration; fractional distillation; neutralization;
oxidation; reduction; thermal decomposition.
REFERENCE SECTION: REDOX POTENTIALS
Chemistry texts are often peppered with statements of the following
type: 'potassium is a strong reducing agent'; 'calcium is a stronger
reducing agent than magnesium'; 'acidified potassium manganate is a
strong oxidizing agent'; and, 'fluorine is a stronger oxidizing agent
than chlorine'. The quantitative basis for these and other related
descriptions is the (continually expanding) set of physical quantities
known as reduction potentials; a critical compilation of these data
is: A. J. Bard et al., Standard Potentials in Aqueous Solution, Marcel
Dekker, New York, 1985. Numerical values of redox potentials are rarely
used in introductory courses, partly because their correct use is quite
complicated: nevertheless, a selected sub-set of values - as shown in
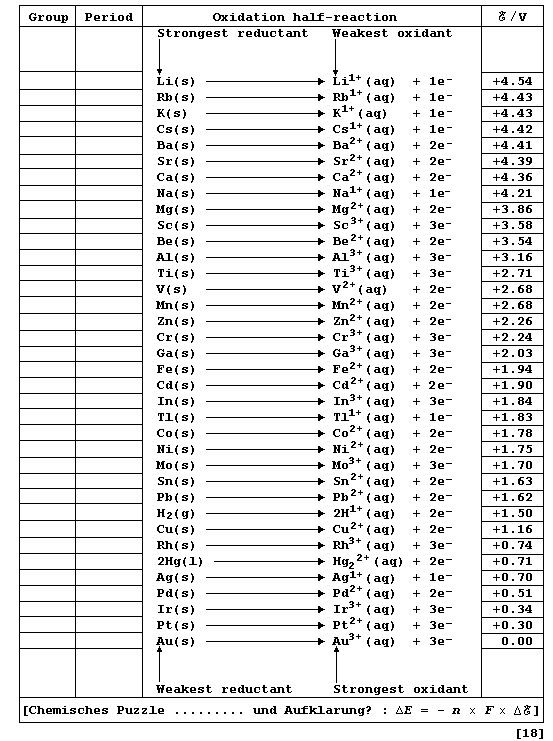
this first Table - does provide a useful point of reference. [Reductant
and reducing agent are synonymous, as are oxidant and oxidizing agent.]

# These data have been obtained, under standardized conditions, using
sophisticated electrochemical cells. The diagram shows the basic design
of such cells; Cell 1 would be suitable for impoverished mortals, but
the well-endowed (or alchemists) would surely prefer Cell 2.

Chemically, a metallic element (M) is characterized by its tendency to
lose one or more electrons and form a positive ion; i.e., it is readily
oxidized (Olé!): M ——————® Mn+ + ne-. One of several different methods
of quantitatively comparing metallic character involves using oxidation
potentials. In principle, these values can be determined by measuring
potential differences of simple electrochemical cells. However, because
published sources of reduction potentials contain data obtained under
standardized conditions, it is both convenient and self-consistent to
derive oxidation potentials from such sources. This derivation involved
changing the sign of each value, assigning an arbitrary value of 0.00 V
to a gold reference electrode, and then executing simple arithmetic.
Complete the Table below, which refers to data obtained under standard
conditions (pH = 0; 298 K), by inserting the group number and period of
each element and by indicating each transition metal with an asterisk.
_______________________________________________________________________
_______________________________________________________________________
Comedy-Dramas for Year 10-12 Students in British Orthography :
[No. 1] [No. 2] [No. 3] [No. 4] [No. 5] [No. 6] [No. 7] [No. 8] [No. 9]
[Dr. Roger Peters : rpeters@wissensdrang.com ; Aufbau1 ; Home Page]